This article was updated July 24, 2020.
Determining how herpes viruses eject their DNA into the nucleus of animal cells could be an important tool to fight the disease. Led by Alex Evilevitch, an associate professor of pathobiology and of physics, the Evilevitch Laboratory, in collaboration with the Microscopy Suite at the Beckman Institute, aims to understand the fundamental aspects of viral infection in order to develop better anti-viral drugs.
Infections caused by herpes viruses can have devastating consequences. This family of viruses infect a range of organisms from humans to chickens. They can damage nerve tissues — including those responsible for vision — or cause cancer. In some cases, there is no treatment. As a result, this family of viruses is one of the most extensively studied, along with HIV and Hepatitis B.
Herpes viruses contain extremely long DNA sequences that are enclosed in a protein shell called the viral capsid. Intriguingly, the DNA is about 500 times longer than the diameter of the capsid. As a result, the DNA needs to be packaged very tightly. When the virus infects a host cell, it travels to the nucleus and ejects its DNA through the nuclear pores. Evilevitch discovered that the pressure with which it does so is 20 atmospheres, which is about 10 times higher than the pressure inside your car tires.
Researchers have studied the steps of viral infection for several years in order to develop better anti-viral drugs. However, the mechanism of DNA ejection still remains poorly understood. “For the past 16 years my lab has worked on how the viral genomes get out of the virus and into the cell,” Evilevitch said.
Before moving to the University of Illinois, Evilevitch was at Carnegie Mellon University, Pittsburgh, for nine years. There he was able to measure the pressure of the viral DNA in the absence of host cells. “After coming to the University of Illinois, we started looking at how this pressure was responsible for infection,” Evilevitch said.
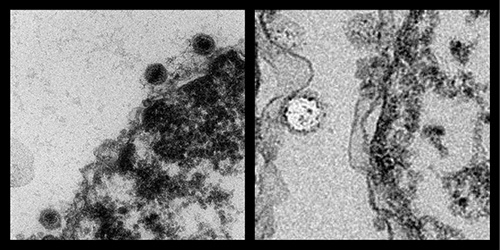
To visualize the DNA ejection from herpes virus into a host cell nucleus, the lab, in collaboration with the Microscopy Suite at the Beckman Institute, makes ultra-thin sections of the viral-infected cells. “We get a cluster of cells which we fix and embed into an epoxy,” said Cate Wallace, a senior microscopist who helps the Evilevitch Lab visualize the cells in the transmission electron microscope. “We cut little pieces of the samples and then make ultra-thin sections of about 90 nm, which is a thousand times smaller than human hair. We set them up in the microscope and find where the herpes viruses are lined up against the nuclear membrane.”
“Beckman, especially Scott Robinson, (manager of the Microscopy Suite), has helped us tremendously in the last year to demonstrate that we can block the DNA ejection,” Evilevitch said.
“The images have been spectacular. We have used them for NIH (National Institutes of Health) proposals and we have published and also submitted a new manuscript using those images,” Evilevitch said. The images have been included in their latest paper “Pressure-driven release of viral genome into a host nucleus is a mechanism leading to herpes infection,” published in eLife, which is the first demonstration of a eukaryotic virus infection driven by DNA pressure.
“The collaboration has been fantastic,” said Alberto Brandariz, a research scientist in the Evilevitch Lab. “I think it is amazing to have this facility on campus.”
Editor's note: An additional paper, "Pressurized DNA state inside herpes capsids—A novel antiviral target," was published in PLOS Pathogens in July 2020. The paper can be found at https://journals.plos.org/plospathogens/article?id=10.1371/journal.ppat.1008604
