Johannes Traa—Artificial Intelligence
“Phase Difference Models and Nonnegative Matrix Factorization for (Audio) Source Separation”
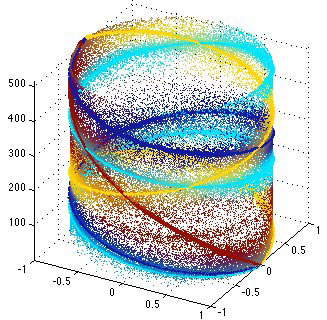
Source separation is a significant problem in the audio realm. Today’s hand-held devices (e.g., cell phones) increasingly have multiple microphones that can be used to improve the quality of the audio extracted from the soundscape around the device. Spatial information is very useful in these contexts and it can be combined with single-channel source modeling techniques like Nonnegative Matrix Factorization (NMF). I will discuss some of my work in this area.
Troy Comi—NeuroTech
“Understanding Biological Complexity--One Cell at a Time”
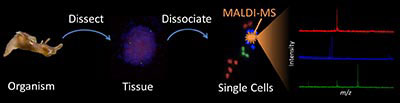
Complex multicellular organisms are composed of a chemically, structurally, and functionally heterogeneous collection of organs, tissues, and ultimately cells. While the analysis of homogenized multicellular structures and cell populations is a well-established approach for biochemical research, recently developed bioanalytical methods offer sufficient sensitivity and precision to accomplish single cell measurements. Single cell analysis is critical for the discovery and investigation of biochemically rare cells including ones involved in pathological and physiological events. By removing the background of neighboring cells, the cellular phenotype may be investigated with minimal interference. We have developed a high-throughput, single cell profiling method based on MALDI mass spectrometry that allows label-free analysis of the peptide contents within dispersed structurally and biochemically heterogeneous cell populations. The method allows the examination of thousands of cells within a single experiment and detects tens to hundreds of distinct molecular features from each cell. From thousands of spectra, we can successfully quantify the relative composition of peptidergic cells from endocrine and nervous tissue.
Jamila Hedhli—Bioimaging Science and Technology
“Molecular Imaging of Stem Cell Induced Angiogenesis at the Onset of Microvascular Complications in Type-1 Diabetes”
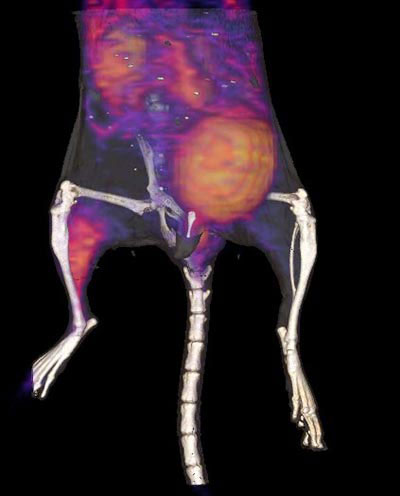
Cardiovascular complications, including peripheral arterial disease (PAD), are among the leading causes of morbidity and mortality in diabetic patients, accounting for over 80 percent of diabetes-associated deaths. Current therapies for patients suffering from PAD rely on mechanical revascularization through either percutaneous or surgical approaches. Unfortunately, these approaches are frequently unsuccessful in the long term. As an alternative, targeted stem cell-based therapies might provide clinical improvement for these patients through improved perfusion by stimulating increased microvascular density (angiogenesis) and subsequent large vessel remodeling (arteriogenesis). Of the various potential candidate cells that might be used in treating PAD patients, mesenchymal stem cells (MSCs) are the most actively pursued at both preclinical and clinical levels. In my talk I will present recent work describing the development of a novel αvβ3-integrin targeted PET-CT tracer (64Cu-PEG4-cRGD2), and its use in evaluating the potential use of muscle-derived mesenchymal stem cells in stimulating angiogenesis and potentially improving muscle function in diabetic mice with hind limb ischemia.